The gastric imprint of microplastic exposure
How microplastics interact with the gut barrier, microbiome, and metabolic signaling
The gastric imprint of microplastic exposure
Plastic pollution is now recognized not only as an environmental problem but also as a biological one. Microplastics and nanoplastics have been detected in human stool, blood, lung tissue, placental tissue, and even arterial plaques. These discoveries confirm that modern plastic exposure does not stop at the environment. It enters the body and interacts with living systems.
One of the first and most important biological environments that ingested microplastics encountered is the intestinal tract. The gut is not simply a digestive tube. It is a complex ecological interface between the outside world and the internal physiology of the body. Its epithelial barrier regulates nutrient absorption, prevents microbial invasion, and communicates with immune and metabolic systems throughout the body. Surrounding and supporting this barrier is the gut microbiome, a dense ecosystem of trillions of microorganisms that influence digestion, immune function, metabolic signaling, and inflammation.
Emerging experimental evidence suggests that microplastics are not biologically inert within this system. Instead, they interact with mucus, epithelial cells, and microbial communities in ways that can alter the structural and functional stability of the gut.
Across multiple experimental systems, several recurring biological patterns have been observed. Microplastic exposure has been associated with thinning of the intestinal mucus layer, reductions in tight junction punctions that maintain barrier integrity, and shifts in microbial populations away from beneficial taxa. These changes frequently coincide with reductions in short chain fatty acids such as butyrate, a key metabolite required for maintaining epithelial health and regulating inflammation.
Taken together, these findings suggest that microplastics may influence the gut ecosystem at several levels simultaneously. They appear capable of affecting physical barrier structures, microbial composition, and metabolic signaling pathways.
While much of the research remains in early stages, the consistency of these patterns across models suggest that microplastics deserve serious consideration as a biological stressor within the gastrointestinal environment.
Entry: microplastics in the digestive system
Human exposure to microplastics occurs through multiple everyday pathways. Food packaging materials, synthetic textiles, tire wear particles, plastic bottles, household dust, and environmental contamination all contribute to a constant background presence of microplastic fragments in modern life.
These particles enter the body primarily through ingestion. Microplastics have been detected in drinking water, seafood, salt, fruits, vegetables, and other foods. Inhaled airborne particles can also become trapped in airway mucus and subsequently swallowed, adding another route of entry into the digestive tract.
Once ingested, these particles travel through the gastrointestinal system. Historically, it was often assumed that most microplastics would simply pass through the digestive tract without meaningful biological interaction. However, growing evidence suggests that the situation is more complex.
Studies have demonstrated that microplastics can interact physically with intestinal mucus and epithelial surfaces. Some particles adhere to the mucus layer that coats the intestinal lining. Others can come into direct contact with epithelial cells or become incorporated into microbial biofilms within the gut environment.
Particle size appears to play a significant role in these interactions. Larger microplastics may remain largely within the intestinal lumen, while smaller particles, particularly nanoplastics, have greater potential to interact with cellular surfaces or penetrate biological barriers.
Evidence of regular ingestion is now well established. Multiple studies have identified microplastics in human stool samples, confirming that plastic particles routinely pass through the gastrointestinal tract. However, laboratory studies indicate that even transient exposure within the gut may be sufficient to trigger biological responses.
Rather than behaving as passive particles, microplastics appear capable of interacting with the complex ecosystem of the gut in ways that may influence both microbial composition and barrier integrity.
Damage to the gut barrier
The intestinal barrier is one of the most remarkable structures in human physiology. Although it is only a single layer of epithelial cells thick, it performs a highly complex task. It must selectively absorb nutrients, electrolytes, and water from digested food while simultaneously preventing harmful bacteria, toxins, and inflammatory molecules from entering the blood stream. Simply put, allow the Good in and keep the Bad out.
This balance is maintained through a coordinated system of structural and biological defenses. A protective mucus layer forms the first physical barrier between microbes and epithelial cells. Beneath this mucus, epithelial cells are connected by tight junction proteins that regulate the permeability of the barrier. Surrounding these structures is the gut microbiome, which helps regulate immune signaling and maintains the stability of the barrier.
Microplastic exposure appears capable of disturbing each of these protective systems.
Experimental studies have observed structural changes in the intestinal barrier following microplastic exposure. These changes include thinning of the mucus layer, reductions in tight junction proteins, and increased permeability of the intestinal lining. When these protective features weaken, the barrier becomes less effective at preventing microbial components and inflammatory molecules from entering circulation.
The result can be a state sometimes described as increased intestinal permeability. Under these conditions, molecules that are normally contained within the gut lumen may begin to cross the epithelial barrier and interact with immune cells in underlying tissues.
A clear illustration of this barrier disruption appears in a recent study published in Nature Communications. In this work, researchers exposed intestinal cell models and mice to polystyrene nanoplastics and observed direct impairment of the gut barrier. Nanoplastic particles were shown to enter intestinal epithelial cells and significantly reduce the expression of key tight junction proteins, including ZO 1 and occludin. Figure 2 of the study provides a visual record of this process. Microscopy images show nanoplastic particles accumulating within enterocyte like Caco 2 cells, followed by visible disruption of the tight junction network that normally seals intestinal cells together. As tight junction proteins declined, permeability of the epithelial layer increased, allowing tracer molecules to pass through the barrier more readily. In other words, the structural “seal” that normally regulates what can pass from the intestinal lumen into the body became measurably compromised. The authors concluded that nanoplastic exposure alters the intestinal microenvironment and directly weakens barrier integrity through changes in host cellular signaling and microRNA regulation.
While the long term implications of these changes remain under investigation, the disruption of barrier integrity is widely recognized as an important factor in inflammatory and metabolic disorders.
Mucus layer disruption
The mucus layer represents the first line of defense within the intestinal barrier. Produced by specialized goblet cells embedded in the intestinal epithelium, this viscous gel forms a physical shield that separates trillions of gut microbes from direct contact with epithelial cells.
This separation is critical. The microbiome contains both beneficial organisms and potentially harmful species. By maintaining distance between microbes and intestinal tissue, the mucus layer allows beneficial microbial activity to occur while limiting inflammatory interactions with host cells.
Research indicates that microplastic exposure may compromise this protective layer.
Several experimental studies have found reductions in mucus production following exposure to microplastic particles. These changes appear to involve both structural and genetic mechanisms. Goblet cell numbers may decrease, and the expression of mucin genes responsible for mucus production can be reduced.
Proteins such as MUC1, MUC2, and MUC3 are central components of intestinal mucus. Reductions in the expression of these proteins lead to a thinner and less protective mucus layer.
When this protective barrier is weakened, bacteria and microbial metabolites can approach epithelial cells more closely. This increased proximity raises the likelihood of immune activation and inflammatory signaling within the intestinal wall.
The mucus layer also interacts closely with certain beneficial microbes. Akkermansia muciniphila, for example, helps maintain healthy mucus turnover and barrier function. Microplastic exposure has been associated with reductions in this bacterium, suggesting a reinforcing cycle in which microbial shifts and mucus depletion may amplify one another.
Tight Junction Disruption and Increased Permeability
Beneath the mucus layer lies the epithelial cell layer that forms the core of the intestinal barrier. These epithelial cells are connected to one another through structures known as tight junctions.
Tight junction proteins function as molecular seals between adjacent cells. They regulate which molecules can pass between cells and enter underlying tissues.
Key proteins involved in this system include claudin 1, occludin, and ZO 1. Together, they form a dynamic gate that maintains the selective permeability of the intestinal barrier.
Experimental studies suggest that microplastic exposure can reduce the expression of these proteins. When tight junction protein levels decline, the barrier becomes more permeable.
Increased permeability allows molecules that normally remain confined within the intestinal lumen to cross the epithelial layer. Among the most biologically significant of these molecules is lipopolysaccharide, a component of the outer membrane of certain bacteria.
Lipopolysaccharide is a powerful immune stimulant. Even small amounts entering the bloodstream can trigger systemic inflammatory signaling.
Chronic low level exposure to lipopolysaccharide has been linked to metabolic disorders, insulin resistance, and inflammatory diseases.
In laboratory models, microplastic exposure has been associated with elevated inflammatory cytokines including TNF alpha, IL 1 beta, and IL 6. At the same time, levels of anti inflammatory cytokines such as IL 10 may decrease.
These shifts suggest that barrier disruption may initiate broader immune responses beyond the gut itself.
Microbiome Disruption
The gut microbiome is a complex ecosystem composed of thousands of microbial species. These microbes perform essential functions in digestion, vitamin synthesis, immune education, and metabolic regulation.
A healthy microbiome maintains a dynamic balance between microbial populations. This balance helps prevent the overgrowth of harmful organisms while supporting the beneficial functions of symbiotic bacteria.
Microplastic exposure has repeatedly been associated with disruptions to this microbial equilibrium.
Studies across multiple animal models have documented reductions in beneficial microbial groups such as Lactobacillus, Bifidobacterium, and Akkermansia. These organisms play important roles in maintaining gut barrier function and regulating immune responses.
At the same time, certain opportunistic or inflammatory species appear to increase in abundance following microplastic exposure.
Examples include Desulfovibrio, Escherichia Shigella, and several Clostridium related taxa. Some of these microbes produce metabolites that can damage epithelial cells or promote inflammation within the gut.
This shift toward a more inflammatory microbial community is commonly referred to as dysbiosis.
Dysbiosis does not simply reflect changes in microbial composition. It often corresponds with alterations in microbial metabolism and signaling. As microbial populations change, the biochemical products they produce also shift.
These metabolic changes can influence host physiology, particularly in relation to immune signaling and metabolic regulation.
Disruption of Short Chain Fatty Acid Metabolism
Among the most important metabolic products generated by the gut microbiome are short chain fatty acids. These molecules are produced when beneficial bacteria ferment dietary fibers that reach the colon.
The primary short chain fatty acids are acetate, propionate, and butyrate.
Butyrate holds particular importance for intestinal health. It serves as the primary energy source for colonocytes, the epithelial cells lining the colon. Adequate butyrate supply supports epithelial repair, maintains tight junction integrity, and helps regulate inflammatory signaling pathways.
Butyrate also plays roles beyond the intestine. It influences immune cell differentiation, metabolic regulation, and even communication between the gut and the brain.
Microplastic exposure has been associated with reductions in bacteria responsible for butyrate production. These include members of the genera Faecalibacterium, Roseburia, and Butyricicoccaceae.
When these microbial populations decline, butyrate production decreases.
Reduced butyrate availability can weaken epithelial cells, making the intestinal barrier more vulnerable to injury and inflammation. In parallel, changes in acetate and propionate levels may alter metabolic signaling related to appetite regulation and hepatic glucose production.
These metabolic changes may represent one of the key pathways through which microplastic induced dysbiosis could influence systemic physiology.
Cellular Stress and Systemic Effects
At the cellular level, microplastics appear capable of inducing oxidative stress and disrupting mitochondrial function.
Mitochondria act as the energy producing structures within cells. When mitochondrial activity is impaired, cells may produce excess reactive oxygen species while generating less usable energy.
This imbalance can damage cellular structures and activate inflammatory pathways.
Experimental models have observed mitochondrial dysfunction in intestinal cells exposed to microplastic particles. These effects are often accompanied by increased oxidative stress markers and elevated inflammatory cytokine expression.
When viewed at the level of whole organs, these cellular changes may contribute to broader metabolic disturbances.
Animal studies have reported liver inflammation, altered lipid metabolism, and abnormal fat accumulation following chronic microplastic exposure.
Although human research is still emerging, these findings suggest that intestinal interactions with microplastics could potentially influence systemic metabolic pathways.
A Converging Pattern of Evidence
Across experimental systems, a consistent biological pattern appears to be emerging.
Microplastic exposure is associated with weakening of the gut’s structural defenses, disruption of beneficial microbial populations, and reductions in microbial metabolites that normally support intestinal health.
At the same time, inflammatory signaling pathways appear to become more active.
These changes create a physiological environment that is more permeable, more metabolically stressed, and more prone to inflammatory signaling.
The gut is one of the body’s most important interfaces with environmental exposures. When disturbances occur within this system, their effects can extend beyond digestion to influence immune regulation, metabolic health, and systemic inflammation.
Although many questions remain unanswered, the existing evidence suggests that microplastics are capable of interacting with biological systems in ways that merit continued scientific investigation.
Breakdown of the gut lining
Mucin reductions
Tight junction decreases
Butyrate reductions
Epithelial cell models
[[ Stuff for later ]]
The gastric imprint of microplastic contamination
How microplastics damage the gut barrier, disrupt the microbiome, and influence whole body health.
Microplastics are not long just “out there” in the environment. They are in us.
These plastics shed from packaging, clothing, tires, and household items. They’ve already been detected in human feces, blood, tissues, and organs confirming that they don’t simply just pass through unchecked.
Once inside the gut, microplastics interact with one of the most important systems in the human body: the gut and its microbiome. This protective filter and dynamic microbial ecosystem support digestion, immune balance, inflammation control, and metabolic health.
And when they disturbed, the effects may ripple past the gut.
Part 1: Ingestion
Every day, we swallow micro- and nanoplastics through the food, water, and air. Once they reach the intestines, they don’t just pass through inertly. Studies show some of them physically interact with gut mucus, epithelial cells, and resident microbes.
What begins as simple digestion becomes a cascade of biological changes.
Part 2: Barrier injury
The gut barrier is only one cell thick, yet it must allow nutrients in while keeping toxins and bacteria out. Microplastics strain this system in three (3) major ways.
1. Damage to tight junctions and a thinner, more fragile mucus layer.
Damage to tight junctions: Nanoplastics have been shown to impair tight-junction proteins (claudin-1, occludin, ZO-1). A 2025 study showed PS nanoplastics significantly increased intestinal permeability. This allows bacterial components such as LPS to enter circulation, triggering chronic, low-grade inflammation.
A 2024 study dosed mice with 3 different amounts of polypropylene microplastics and found that inflammation, weakened barrier and lower mucus levels in a dose dependent manner. Smaller plastics caused more damage than larger particles. [[ Jia 2023 Toxics ]]
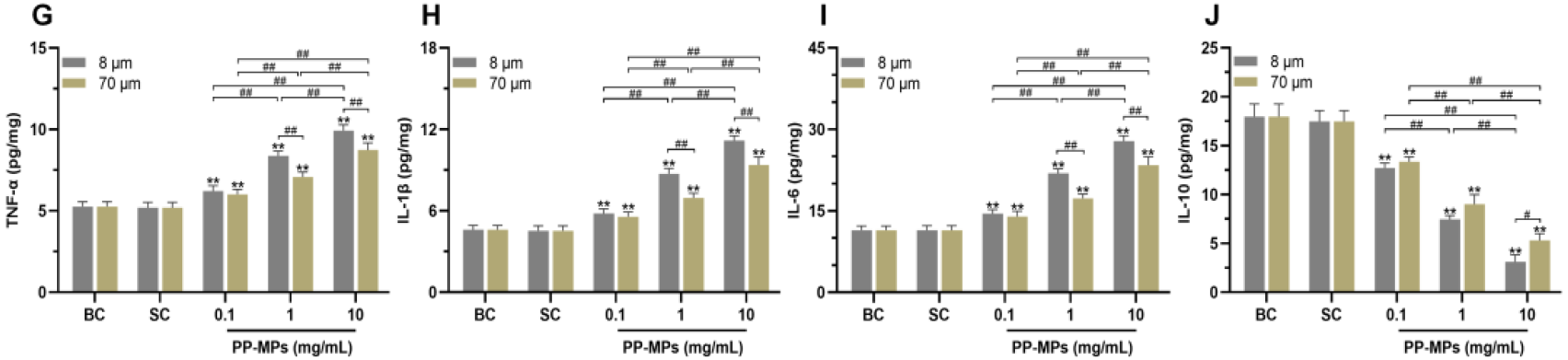
Cells exposed to polypropylene microplastics released much higher amounts of three well-known inflammatory signals: TNF-alpha, IL-1β, and IL-6. At the same time, levels of IL-10, an anti-inflammatory signal, dropped.
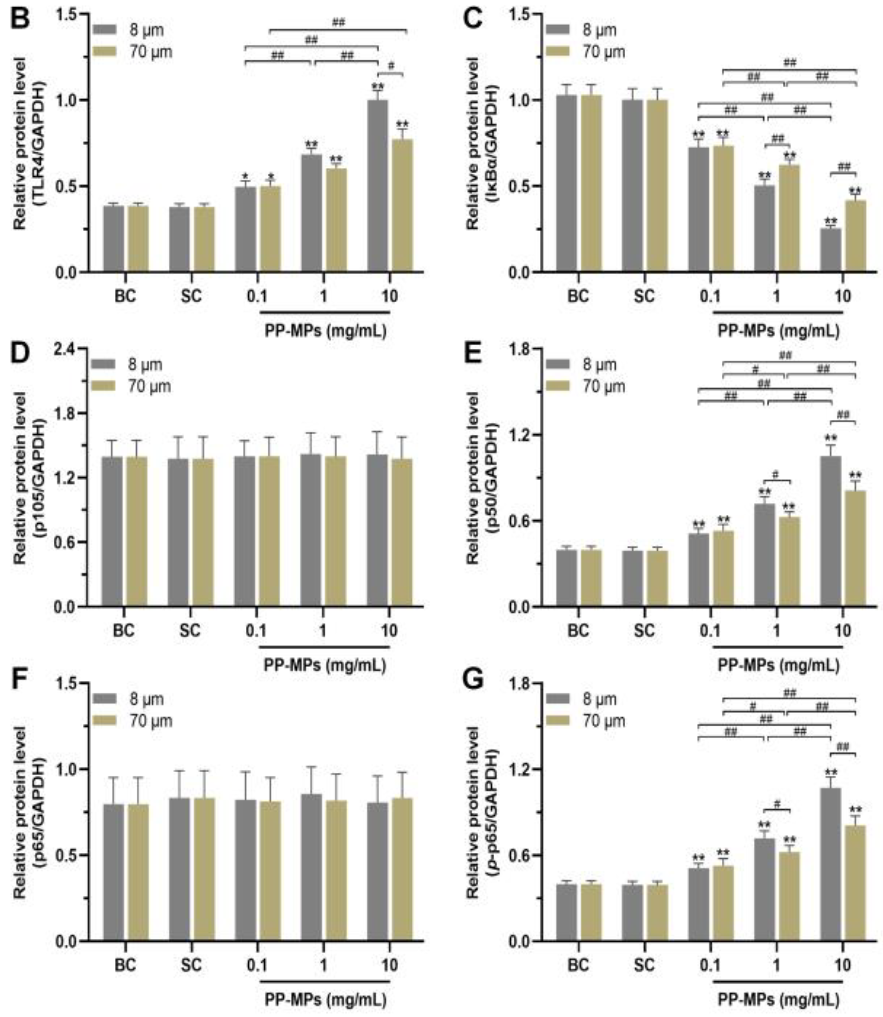
Further, proteins involved in sensing threats and turning on inflammation, TLR4, p50, and p-p65, all increased. Another protein, IκBα, which normally keeps inflammation in check, went down.
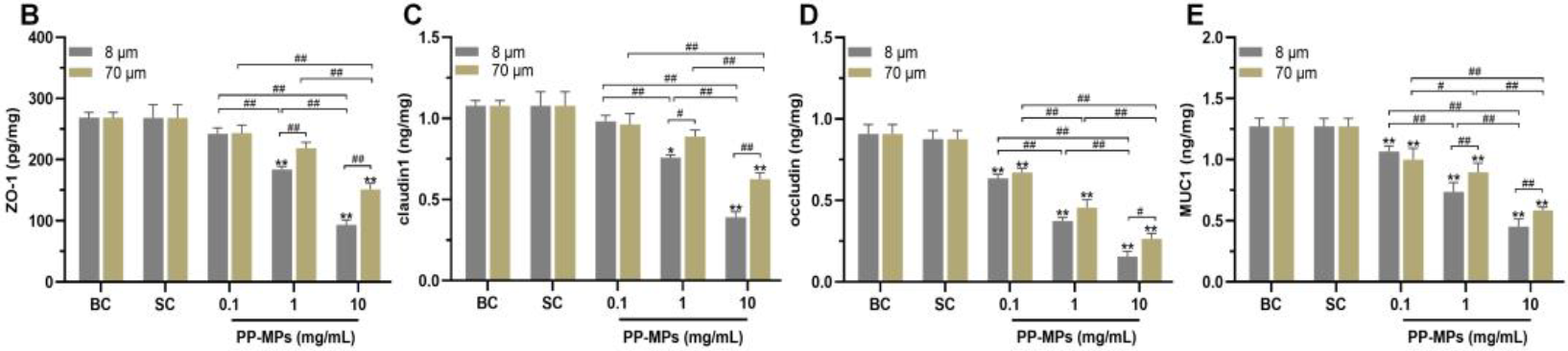
The gut barrier became weaker. Key proteins that help seal the gut lining, occludin, ZO-1, claudin-1, and the mucus-related protein MUC1, all dropped when cells were exposed to microplastics.
The effects were stronger with smaller particles (8 μm) and higher concentrations. This indicates that smaller microplastics do more damage to the gut barrier than larger ones, especially at higher doses.
Across multiple studies, PS exposure has been shown to:
- Impair goblet cells, the cells responsible for producing protective mucus.
- Reduce MUC2 and Muc2 expression, leading to a thinner mucus layer.
- Decrease the number of goblet cells and suppress key genes needed for mucus production (Muc1, Muc2, Muc3) and for goblet cell differentiation (Klf4).
- Lower overall mucus secretion, weakening one of the gut’s most important defense barriers.
[[ Zhai 2024 Environmental Health Perspectives ]] [[ Choi 2021 International Journal of Molecular Sciences ]] [[ Lu 2018 Science of the Total Environment ]] [[ Jin 2019 Science of the Total Environment ]] [[ Wei 2025 Ecotoxicology and Environmental Safety ]]
And that isn’t just the end. Studies have shown that the energy machinery of intestinal cells - the mitochondria - become disrupted when exposed to microplastics. These are the tiny engines that keep cells functioning, repairing, and communicating. Then zoom out to the organ, and the effects become even more visible. In animal studies, microplastic exposure leads to inflammation accumulating in the liver and disruptions in the way fats are processed and stored.
Part 3: Microbiome disruption
Loss of beneficial bacteria
Multiple models have shown reductions in foundation gut taxa
- Akkermansia - key to mucus integrity [[ Agrawal 2024 Science of the Total Environment ]] [[ Wei 2025 Ecotoxicology and Environmental Safety ]]
- Lactobacillus - protective and anti-inflammatory
- Bifidobacterium - crucial for early-life and adult gut homeostasis
- SCFA-products such as Faecalibacterium, Roseburia, Butyricicoccaceae
- Polypropylene and polystyrene particles have been shown to reduce mucus secretion. At the same time, microplastic exposure lowers Akkermansia levels, a bacteria essential for maintaining that mucus layer. A weakened mucus lining allows microbes and toxins to come into closer contact with epithelial cells.
These losses weaken metabolic capacity, immune regulation, and barrier repair.
Rise of opportunistic and inflammatory species
As beneficial bacteria delice, pro-inflammatory, stress-tolerant bacteria expand:
- Desulfovibrio - which produces epithelial-damaging hydrogen sulfide
- Escherichia-Shigella - elevated in both human and mouse microplastic studies
- Parasutterella, Clostridium, and others linked to chronic inflammation.
This shift, in other words dysbiosis, is one of the most consistently observed outcomes across microplastic studies.
Part 4: SCFA disruption
Short-chain fatty acids (SCFAs) are metabolic molecules produced by beneficial bacteria. They serve as fuel and signaling molecules.
When SCFA-producing bacteria drop, so do the levels of SCFAs, including butyrate. Butyrate energizes cells, strengthens tight junctions, supports mucus production, calms inflammation via NF-kB inhibition, and regulates glucose and fat metabolism. Less butyrate leaves the barrier under-powered, inflamed, and more permeable.
Microplastics also have been shown to shift acetate and propionate levels, which are associated with appetite dysregulation and altered hepatic glucose production.
Reduced SCFA availability is one of the clearest mechanistic links between microplastic exposure and downstream metabolic dysfunction.
Part 5: The big picture
Microplastics are reshaping the gut ecosystem in lab, animal, and human studies by reducing beneficial, SCFA-producing bacteria, weakening structural defenses, and increasing inflammation.
Researchers are still uncovering how deeply this cascade runs. But study after study is revealing a consistent pattern. That microplastics are active participants in shaping our biology. Not just passive travelers.
Part 6: Where Winnow fits in
If microplastics erode the gut’s defenses, then strengthening those defenses becomes an important part of the picture.
Winnow was built around two (2) pillars.
- Replacing what modern life depletes. Microplastic exposure is repeatedly associated with reductions in beneficial microbes - especially Bifidobacterium, Lactobacillus, and Lactococcus. These groups play essential roles in barrier support, microbial balance, and the production of short-chain fatty acids. Winnow combines these foundational strains in meaningful amounts, to help supply those beneficial microbes.
- Adding targeted strains shown to bind microplastics in preclinical models. Alongside the foundational strains, Winnow includes a set of bacteria shown in lab and mouse studies to bind common microplastic particles and increase their excretion. This research is still preclinical, but exceptionally encouraging. It highlights the possibility of supporting the gut’s natural clearance pathways rather than relying solely on downstream interventions.
Together, this creates a simple idea:
Rebuild what keeps the gut strong. Add strains that may help defend against modern exposures. Support the system where those exposures first arrive.
Microplastics are now inseparable from modern life. These microscopic fragments shed from packaging, clothing fibers, tiers, and even household items have been detected in the food we eat, the water we drink, and the air we breathe. Their presence in human feces, blood, tissue, and organs confirms what was once only suspected: microplastics do not remain in the environment alone. They enter and accumulate within us.
A growing body of research shows that these particles are not inert materials simply passing through the digestive tract. Instead, they interact directly with the gut microbiome. The dense, dynamic community of trillions of microorganisms that helps regulate digestion, immunity, inflammation, and metabolic health. When this ecosystem is perturbed, the effects can ripple far beyond the gut.
It’s our food, our water. So, yes. It’s in us. So today we’re going to talk about what happens when it gets inside and meets our internal world. So what happens when we swallow microplastics. It sounds like a simple question. But the answer, a complex chain of events.
They tip the balance of the gut microbiome
This is where things get really fascinating. They reshape our internal ecosystem. A healthy gut is all about balance. Microplastics come in and shove that balance out of whack. A thing called dysbiosis. A drop in beneficial bacteria and at the same time a rise in the opportunists. Bacteria that can cause trouble.
One of the most consistent observations across recent studies is that microplastic exposure causes gut dysbiosis, a disruption of the normal microbial equilibrium. This shift involves both a loss of beneficial bacteria and a rise in opportunistic, stress-tolerant, or pro-inflammatory species.
This has been shown in sophisticated lab models, animal models, and observational human studies.
One of these models was used in a 2024 study, to simulate what happens when you introduce plastics into a real human microbial community. There was a stark drop in Akkermanisa, important in helping maintain a healthy mucus lining. Good guys like Bifido and lacto. also took a hit.
One study isn’t enough. But scientific signals keep showing up study after study. These models are being echoed in animal studies and lab models.
For example a 2024 study in mice showed these exact same drops. The same study show a ratio of 2 major groups of bacteria the firmicutes and the bacteriodes get thrown off, which is a red flag often linked to obesity and metabolic issues.
And if you go even simpler, you can see bacteria sticking to plastics in the lab.
Loss of beneficial bacteria
Multiple studies in mice and other models show a reduction in key microbial groups essential for gut health and barrier maintenance:
- Akkermansia: Exposure to PVC microplastics significantly reduced Akkermansia, a mucin-degrading bacteria crucial for gut barrier integrity (Ecotoxicology and Environmental Safety, 2022).
- Lactobacillus: PVC exposure decreases Lactobacillus abundance in mouse models of colitis (Environmental Research, 2025).
- Bifidobacterium: Offspring of polystyrene nanoplastic-exposed mothers showed marked reductions in Bifidobacterium pseudolongum (2024).
- Overall diversity: High PS exposure lowered bacterial richness and diversity, a central marker of microbial resilience (Ecotoxicology and Environmental Safety, 2024).
Diminishing these taxa weakens the gut’s metabolic capacity, immune signaling, and physical defense systems.
The drop in beneficial bacteria has other effects, including on the product of short-chain fatty acids and volatile fatty acids. Metabolites that play central roles in gut and metabolic health.
Butyrate (the gut’s preferred fuel)
Butyrate is produced by bacteria such as Faecalibacterium, Roseburia, and Butyricicoccaceae. It
- Fuels intestinal epithelial cells
- Strengthens tight junctions
- Reduces inflammation via NF-kB inhibition
- Supports mucus production
- Regulates glucose and lipid metabolism
Microplastic exposure reduces SCFA-producing taxa, which means less butyrate. This leaves epithelial cells under-fueled, the barrier weakened, and the immune system more reactive.
Further acetate and propionate help regulate appetite, lipid synthesis, and hepatic glucose production. Dysbiosis caused by MPs and NPs has been associated with shift in acetate:proprionate ratios, patterns often seen with metabolic disorders.
Reduced SCFA availability is one of the clearest links between microplastic-induced dysbiosis and downstream metabolic dysfunction.
Rise in opportunists
As beneficial microbes decline, stress-tolerant and pro-inflammatory species extend. Sometimes dramatically.
- Desulfovibrio: Commonly elevated after PS exposure. It produces hydrogen sulfide (H₂S), which in excess damages epithelial cells and disrupts mucus integrity.
- Escherichia-Shigella: Elevated in PS microplastic-exposed mice and found in higher abundance in preschool children with high fecal microplastics (eBioMedicine, 2023).
- Parasutterella, Colidextribacter, Clostridium sensu stricto 1: Enriched in PVC-exposed mice with colitis; these are associated with chronic inflammation.
Microplastics weaken the gut’s structural defenses
In the intestines, they come up against one of the most important defensive structures in your body, the gut barrier. This barrier is just incredible. It’s like the ultimate fortress wall. But it's only 1 cell thick. And it has to do this constant delicate dance of letting all the good stuff (nutrients and water) while keeping out the bad stuff (toxins and nasty bacteria). But here’s the thing, that wall isn’t invisible.
And this one place where microplastics can cause problems. Animal studies are showing they weaken it in a few key ways. First, they can thin out the protective mucus that can bind the gut. Then they can damage the mortar, these proteins that hold the cellular bricks together. This can literally create a leaky gut. Then they can shrink the projections that help absorb nutrients.
The gut’s two major layers of protection, the mucus lining and the tight junctions between epithelial cells, are physically disrupted by microplastics.
A thinner, vulnerable mucus layer
Studies show that ingestion of PP and PS microplastics reduces mucus secretion, thinning the protective layer that physically separates bacteria from epithelial cells. When paired with decreased Akkermansia (which normally helps maintain this layer), the result is a weakened shield.
A thin mucus layer:
- allows bacteria to come into closer contact with epithelial cells
- increases the risk of microbe-induced inflammation
- reduces the gut’s resilience to dietary and chemical stressors
A “leaky” gut wall
Nanoplastics can impar the integrity of tight junction proteins like Claudin-1, occludin, ZO-1
A 2024 study found that PS nanoplastics lowered the expression of these proteins, leading to increased intestinal permeability.
This “leaky gut” allows microbial components, especially lipopolysaccharide (LPS), to move into the bloodstream. LPS is such a potent immune activator that it is sometimes used in research as a vaccine adjuvant to deliberately stimulate inflammation.
When LPS leaks chronically, the result is systemic, low-grade inflammation, a hallmark of metabolic syndrome, fatty liver disease, and insulin resistance.
Where this leaves us
Microplastics reshare the gut microbiom e in a way that reduces beneficial SCFA-producing bacteria, weakens the gut’s structural defenses, and increases inflammatory signaling. This combination creates a biological environment that is more permeable, more stressed, and more prone to metabolic disruption.
Scientists have already linked dysbiosis to some of the connecting an imbalance gut to some of the biggest metabolic diseases of our time. And the research on microplastics are starting to show how they can be a piece of this puzzle all the way down to the levels of our cells.
What happens to human intestinal cells when they are exposed to microplastics. Disruption to the cell’s energy making factories. Put simply the tiny engines that power our bodies are being messed with.
And here is what that chaos can look like at the cellular level when you zoom out to a whole organ. Inflammation building up in the liver of mice exposed to plastics. And fat metabolism thrown completely out of whack. Front he cell to the organ to the whole system.
And the end result of this, well as multiple studies in animal models have found. Long term exposure to microplstics can lead straight to things like abnormal weight gain and how the body processes fats.